Nama Dosen :
1. Dra. Wiendartun, M.Si
2. Dra. Heni, M.Si.
3. Drs. Yuyu R. Tayubi, M.Si.
Pustaka :
1. Kittel Charles, Introduction to Solid State Physics 7th.ed, 1996, John Wiley & Sons, New York
2. Ashcroft and Mermin, Solid State Physics, 1976, Saunders College , Philadelphia.
Sinar X
-
Merupakan radiasi elektromagnetik berenergi tinggi
-
Dihasilkan akibat interaksi antara berkas berkas elektron eksternal dengan elektron pada kulit atom.
-
Spektrum sinar x memiliki:
panjang gelombang antara10-5-1 nm,
frekuensi antara 1017-1020 Hz,
Energi antara 103-106 eV.
Prinsip difraksi Sinar X
-
Sinar X terpancar dari tabung Sinar X.
-
Difraksi sinar X yang konvergen diterima slit.
-
Sinar X diterima detektor,
diubah menjadi sinyal listrik.
-
Sinyal ini dihitung sebagai analisa pulsa tinggi.
Interaksi Sinar X dengan material
-
Energi berkas Sinar X terserap oleh atom.
-
Energi berkas Sinar X dihamburkan oleh atom
Difraksi Sinar X
- Proses hamburan sinar X oleh bahan kristal.
- Difraksi tergantung pada struktur kristal dan panjang gelombang.
-
- jika (λ) ukuran atom, tidak terjadi difraksi
- jika (λ) < ukuran atom, terjadi difraksi
Difraksi Sinar X
-
Teknik yang digunakan dalam karakterisasi material.
-
Untuk mendapatkan informasi mengenai ukuran atom.
Hukum Bragg
n = 1,2,3,…. orde pertama, kedua, ketiga dst
d jarak antara 2 bidang pantul yang berdekatan
θ sudut antara sinar datang dan sinar pantul
Interferensi konstruktif terjadi jika selisih lintasan antara dua sinar berurutan merupakan kelipatan dari panjang gelombangnya (λ)
Karakterisasi XRD Kristal
7.4 Pertemuan ke -4 : Difraksi sinar- x oleh kristal
7.5 Pertemuan ke -5 : Ikatan Kristal
X-ray crystallography
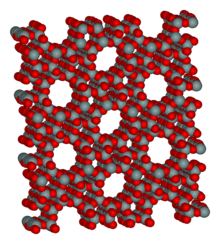
X-ray crystallography is a method of determining the arrangement of atoms within a crystal, in which a beam of X-rays strikes a crystal and diffracts into many specific directions. From the angles and intensities of these diffracted beams, a crystallographer can produce a three-dimensional picture of the density of electrons within the crystal. From this electron density, the mean positions of the atoms in the crystal can be determined, as well as their chemical bonds, their disorder and various other information.
Since many materials can form crystals — such as salts, metals, minerals, semiconductors, as well as various inorganic, organic and biological molecules — X-ray crystallography has been fundamental in the development of many scientific fields. In its first decades of use, this method determined the size of atoms, the lengths and types of chemical bonds, and the atomic-scale differences among various materials, especially minerals and alloys. The method also revealed the structure and functioning of many biological molecules, including vitamins, drugs, proteins and nucleic acids such as DNA. X-ray crystallography is still the chief method for characterizing the atomic structure of new materials and in discerning materials that appear similar by other experiments. X-ray crystal structures can also account for unusual electronic or elastic properties of a material, shed light on chemical interactions and processes, or serve as the basis for designing pharmaceuticals against diseases.
In an X-ray diffraction measurement, a crystal is mounted on a goniometer and gradually rotated while being bombarded with X-rays, producing a diffraction pattern of regularly spaced spots known as reflections. The two-dimensional images taken at different rotations are converted into a three-dimensional model of the density of electrons within the crystal using the mathematical method of Fourier transforms, combined with chemical data known for the sample. Poor resolution (fuzziness) or even errors may result if the crystals are too small, or not uniform enough in their internal makeup.
X-ray crystallography is related to several other methods for determining atomic structures. Similar diffraction patterns can be produced by scattering electrons or neutrons, which are likewise interpreted as a Fourier transform. If single crystals of sufficient size cannot be obtained, various other X-ray methods can be applied to obtain less detailed information; such methods include fiber diffraction, powder diffraction and small-angle X-ray scattering (SAXS). If the material under investigation is only available in the form of nanocrystalline powders or suffers from poor crystallinity, the methods of electron crystallography can be applied for determining the atomic structure.
For all above mentioned X-ray diffraction methods, the scattering is elastic; the scattered X-rays have the same wavelength as the incoming X-ray. By contrast, inelastic X-ray scattering methods are useful in studying excitations of the sample, rather than the distribution of its atoms.
Textbooks
- Blow D (2002). Outline of Crystallography for Biologists. Oxford: Oxford University Press. ISBN 0198510519.
- Burns G., Glazer A M (1990). Space Groups for Scientists and Engineers (2nd ed.). Boston: Academic Press, Inc. ISBN 0121457613.
- Clegg W (1998). Crystal Structure Determination (Oxford Chemistry Primer). Oxford: Oxford University Press. ISBN 0198559011.
- Cullity B.D. (1978). Elements of X-Ray Diffraction (2nd ed.). Reading, Massachusetts: Addison-Wesley Publishing Company. ISBN 0534553966.
- Drenth J (1999). Principles of Protein X-Ray Crystallography. New York: Springer-Verlag. ISBN 0387985875.
- Giacovazzo C et al. (1992). Fundamentals of Crystallography. Oxford: Oxford University Press. ISBN 0198555784.
- Glusker JP, Lewis M, Rossi M (1994). Crystal Structure Analysis for Chemists and Biologists. New York: VCH Publishers. ISBN 0471185434.
- Massa W (2004). Crystal Structure Determination. Berlin: Springer. ISBN 3540206442.
- McPherson A (1999). Crystallization of Biological Macromolecules. Cold Spring Harbor, NY: Cold Spring Harbor Laboratory Press. ISBN 0879696176.
- McPherson A (2003). Introduction to Macromolecular Crystallography. John Wiley & Sons. ISBN 0471251224.
- McRee DE (1993). Practical Protein Crystallography. San Diego: Academic Press. ISBN 0124860508.
- O'Keeffe M, Hyde B G (1996). Crystal Structures; I. Patterns and Symmetry. Washington, DC: Mineralogical Society of America, Monograph Series. ISBN 0939950405.
- Rhodes G (2000). Crystallography Made Crystal Clear. San Diego: Academic Press. ISBN 0125870728., PDF copy of select chapters
- Rupp B (2009). Biomolecular Crystallography: Principles, Practice and Application to Structural Biology. New York: Garland Science. ISBN 0815340818.
- Zachariasen WH (1945). Theory of X-ray Diffraction in Crystals. New York: Dover Publications. LCCN 67-26967.
Applied computational data analysis
- Young, R.A., ed (1993). The Rietveld Method. Oxford: Oxford University Press & International Union of Crystallography. ISBN 0198555776.
Tutorials
- Crystallography for beginners
- Simple, non technical introduction
- "Small Molecule Crystalization" (PDF) at Illinois Institute of Technology website
- International Union of Crystallography
- Crystallography 101
- Interactive structure factor tutorial, demonstrating properties of the diffraction pattern of a 2D crystal.
- Picturebook of Fourier Transforms, illustrating the relationship between crystal and diffraction pattern in 2D.
- Lecture notes on X-ray crystallography and structure determination
- Online lecture on Modern X-ray Scattering Methods for Nanoscale Materials Analysis by Richard J. Matyi
Disusun Ulang Oleh:
Arip Nurahman
Department of Physics, Indonesia University of Education
&
Follower Open Course Ware at MIT-Harvard University, Cambridge.USA.
Semoga Bermanfaat dan Terima Kasih
Lihat Juga
Pendahuluan Fisika Zat Padat





Tidak ada komentar:
Posting Komentar